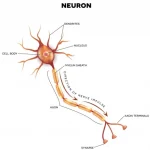
Dr. Alois Alzheimer was a German psychologist and neuropathologist. In 1906 Dr. Alzheimer noticed changes in the brain tissue of his patient, Augusta Deter who had died of an unusual mental illness. Her symptoms included memory loss, language problems and unpredictable behavior. After she died he examined her brain tissue under a microscope and used special stains and found clumps (now called amyloid Plaques) and tangled bundles of fibers, (now called Tau).
These proteins build up slowly over decades and cause previous healthy neurons to stop functioning, lose connections with other neurons and die.
The abnormal build up of these proteins cause changes in the brain that begins about 20-30 years before symptoms appear.